Τη διαδικασία τροποποίησης του σκευάσματος, για όσους έχουν κλείσει ραντεβού για την πρώτη δόση με το εμβόλιο της AstraZeneca διευκρινίζει με ανακοίνωσή της η Εθνική Επιτροπή Εμβολιασμού.
Όπως ανακοινώθηκε κατά την ενημέρωση του υπουργείου Υγείας, τη Δευτέρα, η σύσταση για το εμβόλιο της AstraZeneca τροποποιείται και πλέον θα χορηγείται μόνο σε άτομα άνω των 60 ετών.
Στην παρούσα φάση που στη χώρα μας τα επίπεδα διασποράς του κορωνοϊού είναι μέτρια προς χαμηλά και έχουν σταθερή τάση για περαιτέρω μείωση, η Εθνική Επιτροπή Εμβολιασμών συνιστά τον εμβολιασμό με την 1η δόση του εμβολίου της AstraZeneca για τα άτομα 60 ετών και άνω, στα οποία το όφελος από τον εμβολιασμό υπερτερεί σαφώς του κινδύνου από θρόμβωση με θρομβοπενία που μπορεί να εκδηλωθεί μετά από αυτόν.
Με SMS θα ειδοποιούνται η ενδιαφερόμενοι για την αλλαγή εμβολίου
Με βάση αυτά, όσοι έχουν προγραμματισμένο ραντεβού πρώτης δόσης του AstraZeneca ηλικίας κάτω των 60 ετών, θα λάβουν ένα ενημερωτικό sms για την απόφαση της Επιτροπής και τη σύσταση, αν θέλουν να αλλάξουν εμβόλιο.
Σημειώνεται ότι όσα άτομα 30–59 ετών επιθυμούν να ξεκινήσουν τον εμβολιασμό τους (1η δόση) με το εμβόλιο της AstraZeneca επειδή εκτιμούν ότι έχουν υψηλό ή μέτριο κίνδυνο νόσησης με Covid-19 λόγω των συνθηκών και του τρόπου ζωής τους ή/και προκρίνουν τον ταχύτερο εμβολιασμό τους, θα είναι δυνατό να επιλέξουν το εμβόλιο αυτό.
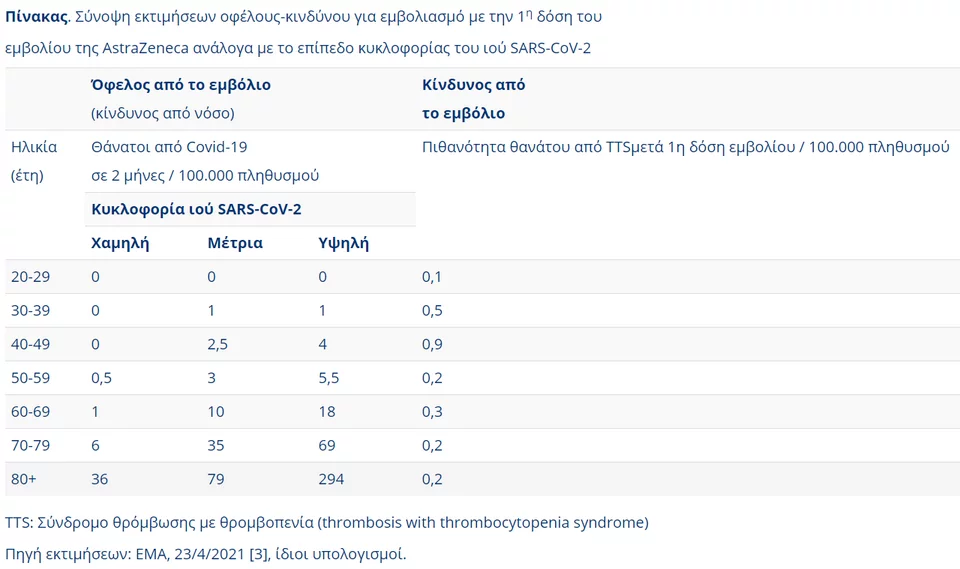
Ο πίνακας κινδύνου εμφάνισης θρομβώσεων μετά από εμβόλιο της AstraZeneca
Μάλιστα, η ανακοίνωση δίνει έναν αναλυτικό πίνακα με την εκτίμηση του κινδύνου από τις διαπιστωθείσες παρενέργειες του εμβολίου σε σύγκριση με τον κίνδυνο βαριάς νόσησης από τον κορωνοϊό ή και θανάτου, ανά ηλικία.
Ποιες κατηγορίες πολιτών μπορούν να αλλάξουν τη δεύτερη δόση του εμβολίου
Όπως ανακοινώθηκε και χθες, άτομα που έχουν ήδη πραγματοποιήσει την πρώτη δόση του εμβολίου της AstraZeneca, ανεξαρτήτως ηλικίας, συνίσταται να συνεχίσουν κανονικά τη 2η δόση με το ίδιο εμβόλιο.
Από τη σύσταση αυτή εξαιρούνται:
1.Άτομα όλων των ηλικιών που κατά το διάστημα των πρώτων εβδομάδων μετά την 1η δόση εκδήλωσαν σοβαρή ανεπιθύμητη ενέργεια θρόμβωσης, με ή χωρίς θρομβοπενία. Η 2η δόση θα πρέπει να γίνει με άλλο εμβόλιο. Στις περιπτώσεις αυτές γίνεται σχετικό αίτημα από θεράποντα ιατρό προς την αρμόδια επιτροπή (μέσω του emvolio.gov.gr).
2.Άτομα όλων των ηλικιών που έχουν άλλον ειδικό λόγο για να γίνει η 2η δόση του εμβολιασμού τους με άλλο εμβόλιο. Θα είναι δυνατόν να γίνει αντικατάσταση του εμβολίου της 2ης δόσης μετά από έγκριση σχετικού αιτιολογημένου αιτήματος από θεράποντα ιατρό προς την αρμόδια επιτροπή (μέσω του emvolio.gov.gr).
3.Άτομα ηλικίας μικρότερης από 30 ετών τα οποία για οποιοδήποτε λόγο έκαναν την 1η δόση εμβολιασμού με το εμβόλιο της AstraZeneca. Συνιστάται η 2η δόση να γίνει με άλλο εμβόλιο, εκτός αν, για λόγους ταχύτερης ανοσοποίησης ή άλλους λόγους, επιθυμούν να συνεχίσουν με τη 2η δόση του εμβολίου της AstraZeneca.
Στις παραπάνω περιπτώσεις θα επιλέγεται κάποιο από τα διαθέσιμα mRNA εμβόλια της Pfizer ή της Moderna.
Τι γνωρίζουμε για το μικτό σχήμα εμβολιασμού
Σημειώνεται από την Επιτροπή ότι δεν έχει τεκμηριωθεί ακόμα η αποτελεσματικότητα του μικτού σχήματος εμβολιασμού (πρώτη δόση με το εμβόλιο της AstraZeneca, δεύτερη δόση με εμβόλιο τεχνολογίας mRNA, δηλ. εμβόλιο της Moderna ή της Pfizer) ως προς την προστασία που παρέχει από τη νόσο Covid-19.
Παρόλα αυτά, η αποτελεσματικότητα του σχήματος για την πρόληψη της νόσου πιθανολογείται βάσιμα καθώς υπάρχουν ευρήματα μελετών που δείχνουν ισχυρή ανοσιακή απόκριση από το μικτό σχήμα, όλα τα εμβόλια –ανεξαρτήτως τεχνολογίας– έχουν ως στόχο την «πρωτεΐνη της ακίδας» του ιού και μικτά σχήματα έχουν δειχθεί αποτελεσματικά στην περίπτωση άλλων εμβολίων.
Θα πρέπει επίσης να αναφερθεί ότι ο εμβολιασμός με το μικτό σχήμα (πρώτη δόση με το εμβόλιο της AstraZeneca, δεύτερη δόση με εμβόλιο mRNA) προκαλεί συχνότερα ήπιες ή μέτριας βαρύτητας αντιδράσεις μετά τη 2η δόση (π.χ. πόνος στη θέση της ένεσης, πυρετός, πονοκέφαλος, κόπωση κλπ.). Ορισμένες Ευρωπαϊκές χώρες έχουν ήδη υιοθετήσει το μικτό σχήμα μετά από πρώτη δόση με το εμβόλιο της AstraZeneca
Πηγή: iefimerida.gr